Three things to know before vaccinating your kids
The Pfizer vaccine was tested for a short period of time on a relatively small number of kids.
All,
I just wanted to get a note out to friends and family as it pertains to the upcoming Pfizer vaccine for use on kids ages 5 to 12. Over the weekend, the FDA released its briefing document discussing the results that Pfizer and BioNTech provided them. This is in preparation for the FDA committee meeting on Oct 26, 2021 to approve Pfizer’s COVID-19 vaccine on young kids. All things considered, it is a small study (less than 2,400 kids at the outset) from which a lot of decisions will be made.
Given the tone of the briefing document and the fact that the US government has announced plans to dose 28 million young kids, it is highly likely this will be approved. Also likely is that it will be available to all kids 5 to <12 years of age, and mandated for many through school boards (public and private) as was the case of school kids from 12 to 17 early this year.
My goal is to stay away from politics or editorializing. I want to limit my comments to factual statements available from the briefing document and other relevant scientific studies. You decide what to do with this information. Most headlines are promoting >90% effectiveness without any worse side-effects than have been seen to date. This is true but it fails to take critical look at what was tested/measured, what was assumed/inferred, and what is different between adults and kids. As parents, we can’t make informed decisions without this information.
What we know about the Pfizer vaccine tested on young kids falls into three main points:
Effectiveness is high (90.7%) but is already waning by the 3rd month. Expect boosters.
Pfizer tested their vaccine on 2,379 kids between June 2021 and early October 2021. Roughly 2/3 of the participants (1,591) were placed in the vaccinated group and 1/3 of the participants (788) were given a placebo to form a control group. For a variety of reasons 1,910 completed the study and were counted towards the vaccine effectiveness calculation. Over the course of four months, 19 kids got COVID-19. Three of these infections were in the vaccinated group. Sixteen were in the control group. Based on this an effectiveness was fairly calculated to be 90.7%. No kids in either group were hospitalized or died.
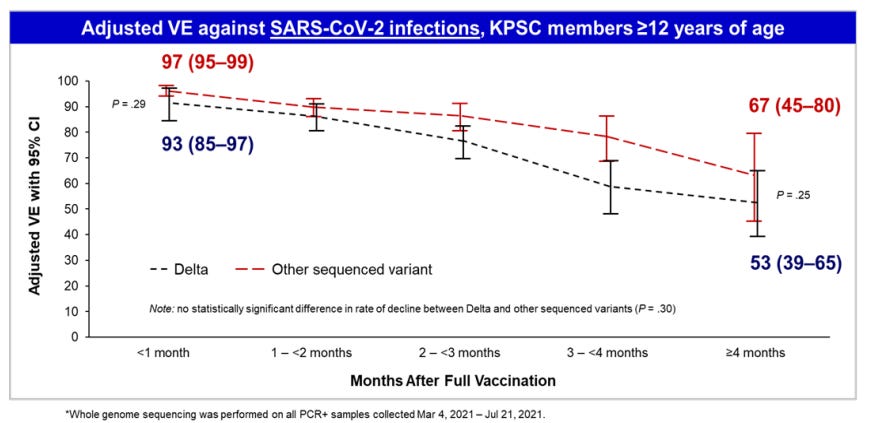
Given the data presented, there is no reason to doubt that vaccine is 90.7% effective at protecting children from getting COVID-19. However, kids were only tracked for a maximum of three months. This occurred during a time when the Delta variant was the dominant strain of the virus so the initial protection seems strong. But we know from the booster discussion that vaccine effectiveness is waning. Pfizer’s own study claimed that effectiveness dropped to 47% after 5 months. Other data has this dropping faster.
The briefing document is silent on this issue. There is no reference to protection over time. However, within the data provided, you can see a waning of effectiveness. The number of cases in the first two months was one (1) in the vaccinated group versus 13 in the unvaccinated control group (96.2% protection – very good). The second month fell right at the height of the Delta surge in the US. But in the third month, two (2) cases for the vaccinated group versus three (3) in the control group. If you look just at the third month, this works out to 67.1% protection using the same calculation method. Therefore, the data is telling us that by the third month, the protection has dropped by a third already. There is every reason to expect that boosters will be promoted six months from now.
Thus 90.7%, while a very good number, is an average of even better protection for two months followed by declining protection by month three. Of course, Pfizer just measured a few handful of cases total out of less than 2,000 kids. We should be careful reading too much into this. Yet that is what the data shows, and it is perfectly consistent with the data Pfizer submitted last month to the FDA when justifying boosters (note month 3 on the Delta curve is just about at 67%):
The vaccine probably does provide good protection in terms of hospitalization, but this was never measured.
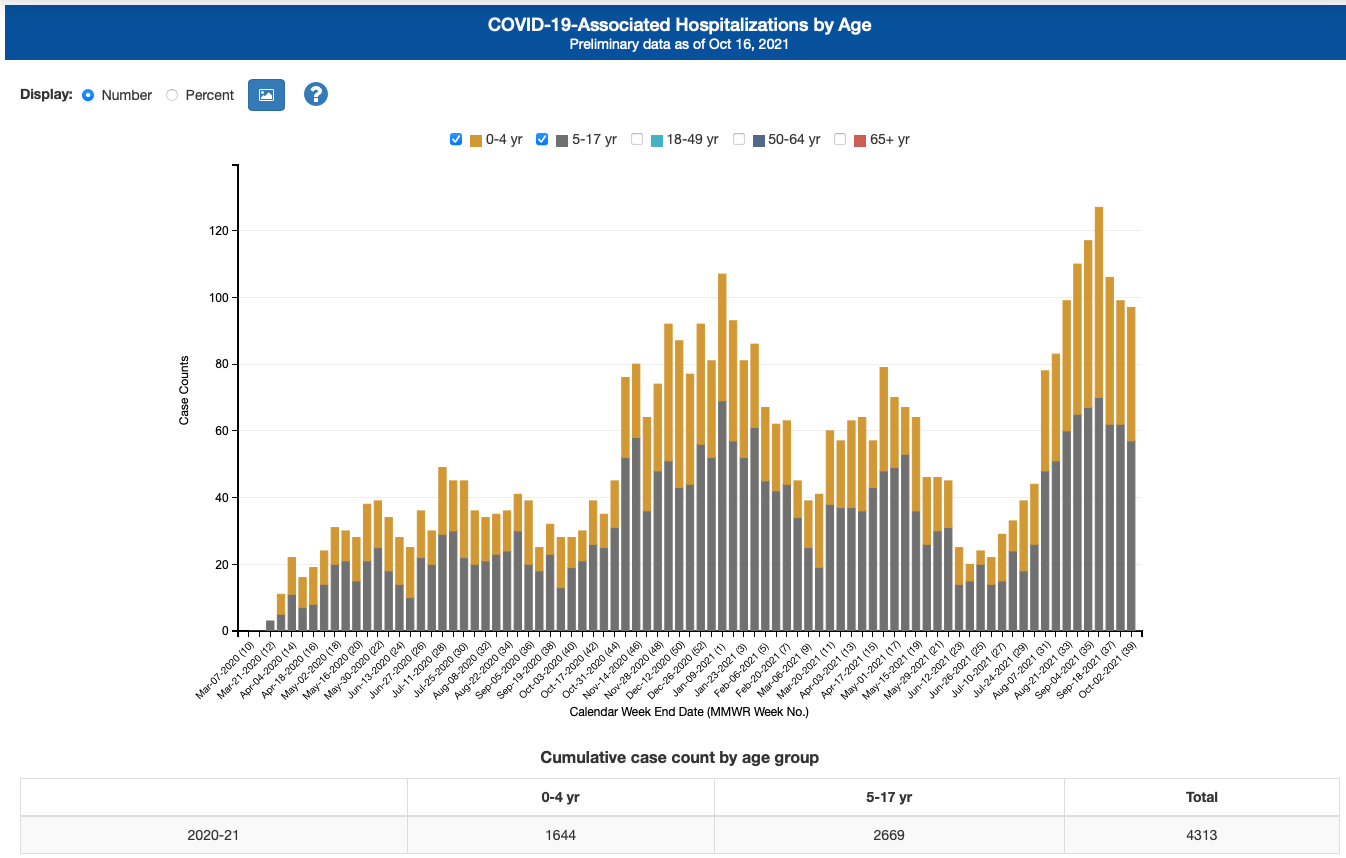
In recent months, the US government and Pfizer have been progressively acknowledging that vaccine effectiveness wanes. Most people have taken comfort in the fact that vaccines still prevent hospitalization and deaths. For good reason, the data still shows this for adults. However, one would think, you’d want to measure this if this has become your primary value. The problem is, you can’t easily measure it in kids, and Pfizer didn’t. Childhood hospitalizations from COVID are just too rare for them to do so. According to the latest CDC posted data there have been 4,313 COVID-hospitalized patients under the age of 17 over the 81 weeks of the pandemic (about 53 per week nationwide):
During the Pfizer trial just 1% of the kids caught COVID-19 and of those the likelihood of any one of them ending up in the hospital was 0.5%. Therefore, it would take clinical trial of more than 100,000 kids to measure a handful of kids (5) that statistically should have been hospitalized. It wasn’t at all practical to measure this. So, what they did was look at the antibodies in the blood, do some math and extrapolate:
- We know 30 microgram of vaccine induces X level of antibodies in the blood in adults.
- Prove that 10 microgram of vaccine induces that same X level of antibodies in kids.
- Now we know adults had >90% protection initially from hospitalization.
- Therefore, children must have the same level of protection from hospitalization.
This is called immunobridging and is common when you don’t have enough data to prove what you want to prove about a vaccine. I am not saying this is wrong. I am just saying that COVID has proven to be different in kids than adults. Just know that the Pfizer study on kids measured nothing as it pertains to hospitalization other than no one (vaccinated or unvaccinated) got sick enough from COVID to go to the hospital. Ask yourself, if COVID hospitalization of kids is so rare that it is not possible to study it in a reasonable amount of time, how necessary is it to vaccinate young kids? I am not suggesting one answer over the other. I am simply saying, this fact begs the question.
So when you see the headline: “CDC study: Pfizer vaccine is 93% effective against COVID-19 hospitalization in youths” you now know they didn’t actually measure this in the study. They simply calculated based on assumptions.
Neither Pfizer nor the FDA have done anything to address the most concerning side effect: Myocarditis in boys and young men. It is more wait and see.
Myocarditis (the inflammation of the heart) is not the only serious side effect. The NIH is also looking into how mRNA vaccines cause menstrual irregularities in women. But Myocarditis is an issue recognized by the CDC and is probably the highest profile side-effect, particularly for younger vaccine recipients. The original clinical trials failed to highlight this risk to the public when seeking initial approval back in December 2020. It has only been because of independent researchers and clinicians using the Vaccine Adverse Events Reporting Systems (https://openvaers.com/covid-data) database that this issue has gained attention. By June 2021 (the start of this Pfizer study in kids), it was openly recognized as a concern.
In the study, Pfizer saw no occurrence of Myocarditis in their study on 5-12 year old kids but they admitted that the sample size of less than 1,000 boys was too small to expect to see it. Their plan is to monitor this as the vaccine is rolled out to millions of kids in the United States and across the globe. In their words:
“The size of the safety database is not large enough to detect any potential risks of myocarditis associated with vaccination. For this reason, long-term safety of the COVID-19 vaccine in participants 5 to <12 years of age will be studied in 5 post-authorization safety studies, including a 5-year follow-up study to evaluate long-term sequelae of post-vaccination myocarditis/pericarditis.”
I have no idea how critical the Myocarditis issue is but given the prevalence in younger people one would think it would have been part of this study. Unfortunately, neither Pfizer nor the FDA are the authority on this issue. Rather than study this critical issue themselves, Pfizer acknowledged the authority Israel has become on this issue. Pfizer/FDA referenced an Israeli study in the New England Journal of Medicine (NEJM) in the briefing confirming that vaccine-induced Myocarditis is most noticeable in young men (16-19). In this demographic, it can occur 15 people out of 100,000 (1 in 6,667). Other researchers suggest it is even more prevalent, but it is safe to assume it happens in at least one in 6,000 young men after their second dose. (Note: this issue is even more prevalent in the Moderna vaccine, likely due to its higher dosage)
Essentially Pfizer is saying, “Based on the dosage is lower and what Israel is telling the world, it should be fine.” Their actual words:
“Israeli safety surveillance databases suggest that incidence rates of rare post-vaccination myocarditis peaks in individuals 16 to 19 years of age (primarily in males) and declines in adolescents 12 to 15 years of age. In addition, the dose for children 5 to <12 years of age is 1/3 of the dose given to older vaccines (10 µg vs. 30 µg).”
Since this work just completed a few weeks ago and the kids were watched for 3 months, we know very little about the vaccine’s long term effect on kids. This was not studied.
Other basic details of the study:
Officially the vaccine is known as BNT162b2 (Pfizer is working to brand this drug as ‘COMIRNATY’) but we all know it as “the Pfizer vaccine.” There is no difference between the vaccine for kids and adults except the dosage. For adults and kids down to 12 years of age, the dosage is 30 micrograms. Pfizer tested and is recommending a 1/3 dosage of 10 micrograms for kids older than 5 and younger than 12. In all cases, two doses taken three weeks apart are required to be considered fully vaccinated.
Normal, expected side effects included pain, swelling or redness at the injection site. This occurred in ¾ of the vaccinated kids. This was mostly mild pain lingering for a few days. The placebo group felt pain at the injection site less than 1/3 of the time.
Serious side effects (a “related adverse event” in clinical trial jargon) were 55 in the vaccinated group and 14 of the placebo group. The most serious side effects were akin to an allergic reaction, treatable with Benadryl or something similar.
Final thoughts
I am not criticizing Pfizer offering the vaccine to kids. Much of the country wants this vaccine for their kids. But parents should know as much as possible. Pfizer found a lower dosage (10 micrograms) that works on kids about as well as 30 micrograms works on adults. This makes sense based on a lower body weight if for no other reason. While Pfizer and the FDA are silent on it, the effectiveness wanes quickly (drops to 67% by month 3) just like what we now see in adults. Serious side effects (ones that could put your child in the hospital) that we know of are exceedingly rare (15 in 100,000) but so is hospitalization from COVID for kids. CDC’s data of 4,313 hospitalized kids ever hospitalized for COVID out of a population of 73 million kids works out to 6 in 100,000.
There is a lot we don’t know about these drugs because there hasn’t been enough time nor enough people studied. The benefit-risk analysis is rather different from that of older people as evidenced by the fact that COVID hospitalizations of kids is so rare. That is why Pfizer didn't see any in the study (even in the unvaccinated) and wouldn’t have expected to see any.
Good luck in making decisions for your family. I hope this helps. Complaints, praises, critiques and other feedback all welcome. Send it bbanish@gmail.com. Time permitting, I’d like send out more important information. If you find this interesting let me know. Forward this to whomever you want.
- Bryan